2017/03/02
Third-generation inhibitors targeting EGFR T790M mutation in advanced non-small cell lung cancer
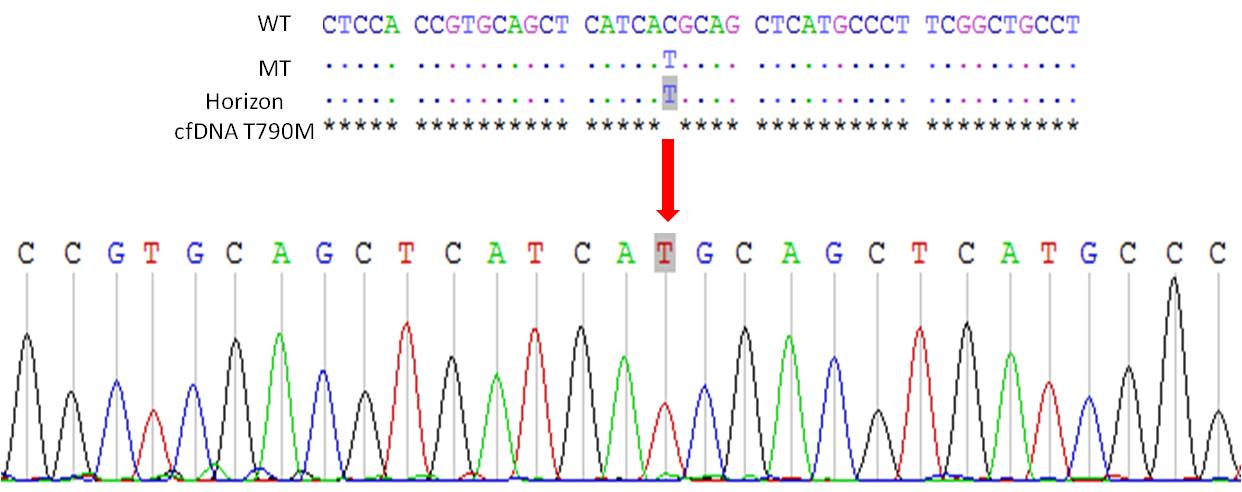
For third-generation inhibitor, limit of detection of EGFR T790M mutation must be reaching 0.1 to 0.01% .
Femtopath now offer you more affordable and high sensitivity T790M mutation kit, especially in plasma cfDNA.
<Abstract>
- The tyrosine kinase inhibitors (TKI) against epidermal growth factor receptor (EGFR) are widely used in patients with non-small cell lung cancer (NSCLC). However, EGFR T790M mutation leads to resistance to most clinically available EGFR TKIs. Third-generation EGFR TKIs against the T790M mutation have been in active clinical development. These agents include osimertinib, rociletinib, HM61713, ASP8273, EGF816, and PF-06747775. Osimertinib and rociletinib have shown clinical efficacy in phase I/II trials in patients who had acquired resistance to first- or second-generation TKIs. Osimertinib (AZD9291, TAGRISSO) was recently approved by FDA for metastatic EGFR T790M mutation-positive NSCLC. HM61713, ASP8237, EGF816, and PF-06747775 are still in early clinical development. This article reviews the emerging data regarding third-generation agents against EGFR T790M mutation in the treatment of patients with advanced NSCLC.
<Reference>
Wang et al. Journal of Hematology & Oncology (2016) 9:34